Discover a World of Materials at Your Fingertips.
Azmater.com is your comprehensive resource for information on a wide variety of materials. Whether you're researching metals, polymers, ceramics, or composites, our website brings you reliable data and insightful details. Explore expertly curated content to support your projects, studies, or professional needs.
Classification of Engineering Materials
Engineering materials are typically categorized into four main groups based on their chemical composition and atomic structure. Each group possesses distinct physical and mechanical properties suitable for specific applications.
| Category | Key Characteristics | Common Examples |
|---|---|---|
| Metals | High thermal and electrical conductivity, ductility, and strength. | Steel, Aluminum, Copper, Titanium. |
| Ceramics | High melting points, brittle, excellent insulators, chemically inert. | Glass, Alumina, Silicon Carbide. |
| Polymers | Low density, flexible, easy to mold, generally non-conductive. | Polyethylene, PVC, Nylon, Rubber. |
| Composites | Combination of two or more materials to achieve superior properties. | Fiberglass, Carbon Fiber, Concrete. |
Trending
Mechanical Properties of Materials
Understanding how materials respond to external forces is crucial for structural design. The following properties define a material's behavior under load:
- Strength: The ability to withstand an applied load without failure or plastic deformation.
- Hardness: Resistance to localized surface deformation or scratching.
- Ductility: The degree to which a material can sustain plastic deformation under tensile stress before failure (e.g., stretching into a wire).
- Toughness: The ability of a material to absorb energy and deform plastically before fracturing.
- Elasticity: The ability of a body to resist a distorting influence and to return to its original size and shape when that influence or force is removed.
The Role of Atomic Bonding
The macroscopic properties of a material are a direct result of the bonds between its atoms. There are three primary types of strong (primary) chemical bonds:
- Metallic Bonding:
Characterized by a "sea of delocalized electrons." This explains why metals are excellent conductors of electricity and heat.
- Ionic Bonding:
Formed by the electrostatic attraction between oppositely charged ions. This results in the hard, brittle nature of most ceramics.
- Covalent Bonding:
Involves the sharing of electron pairs between atoms. This creates extremely strong bonds, as seen in diamonds and silicon.
Thermal Properties and Heat Treatment
Materials react differently to temperature changes, which can be manipulated to change their internal structure. Key concepts include:
Thermal Expansion: Most materials expand when heated and contract when cooled. This is measured by the Coefficient of Thermal Expansion (CTE).
Heat Treatment Processes:
- Annealing: Heating and slow cooling to soften a material and improve ductility.
- Quenching: Rapid cooling to increase hardness (common in steel).
- Tempering: Reheating a quenched material to reduce brittleness while maintaining strength.
Smart and Advanced Materials
Modern engineering has developed "Smart Materials" that can change their properties in response to external stimuli. These are revolutionizing industries from aerospace to medicine.
| Material Type | Stimulus & Response |
|---|---|
| Shape Memory Alloys | Changes shape back to a pre-defined form when heated. |
| Piezoelectric Materials | Produces an electric voltage when mechanical stress is applied (and vice-versa). |
| Photochromic Materials | Changes color or transparency when exposed to light (UV radiation). |
| Thermoelectric Materials | Converts temperature differences directly into electric voltage. |

From basic properties to advanced characteristics, Azmater.com empowers you to find the material information you need quickly and easily. Our user-friendly platform is designed to simplify your search and expand your understanding. No matter your field or application, we connect you with the essential material knowledge that drives innovation.
 azmater.com
azmater.com 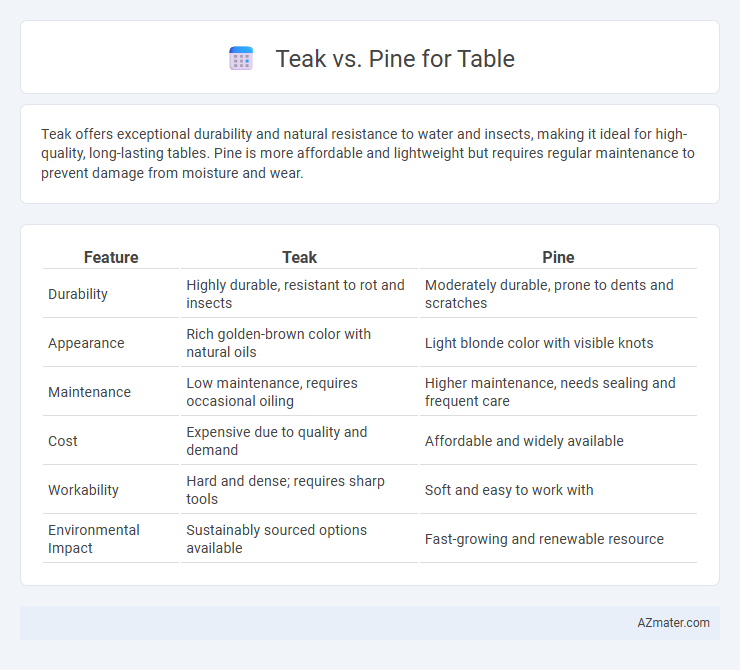 Teak vs. Pine for Table
Teak vs. Pine for Table